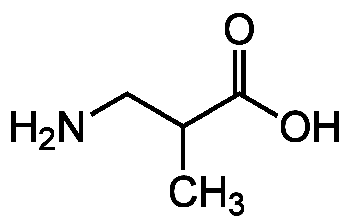
Chemical Structure
3-Aminoisobutyric acid [BAIBA]
AG-CR1-3596
CAS Number1214139-20-5
Product group Chemicals
Estimated Purity>98%
Molecular Weight103.1 . 18.0
Overview
- SupplierAdipoGen Life Sciences
- Product Name3-Aminoisobutyric acid [BAIBA]
- Delivery Days Customer10
- CAS Number1214139-20-5
- CertificationResearch Use Only
- Estimated Purity>98%
- Molecular FormulaC4H9NO2 . H2O
- Molecular Weight103.1 . 18.0
- Scientific DescriptionCatabolite of thymine [1-4]. Precursor for 3-Hydroxyisobutyric acid synthesis [3]. Small molecule myokine. Browning inducer of white adipose tissue (WAT) into brown adipose tissue (BAT) [9, 10]. PGC-1alpha-mediated and exercise-triggered nonadrenergic activator of the thermogenic program in WAT. Increases the expression of brown adipocyte-specific genes (browning) in white adipocytes and beta-oxidation in hepatocytes through a PPARalpha-mediated mechanism [9, 10]. Induces a brown adipose-like phenotype in human pluripotent stem cells and improves glucose homeostasis [9, 10]. Enhances fatty acid oxidation and reduces body weight in mice through an increased production of leptin by the white adipose tissue (WAT) [5-7]. Inhibits Agxt2-mediated metabolism of dimethylarginines [8]. Partial agonist of the glycine receptor (GlyR) [4]. - Chemical. CAS: 214139-20-5 and 144-90-1 (anhydrous). Formula: C4H9NO2 . H2O. MW: 103.1 . 18.0. Catabolite of thymine. Precursor for 3-Hydroxyisobutyric acid synthesis. Small molecule myokine. Browning inducer of white adipose tissue (WAT) into brown adipose tissue (BAT). PGC-1alpha-mediated and exercise-triggered nonadrenergic activator of the thermogenic program in WAT. Increases the expression of brown adipocyte-specific genes (browning) in white adipocytes and beta-oxidation in hepatocytes through a PPARalpha-mediated mechanism. Induces a brown adipose-like phenotype in human pluripotent stem cells and improves glucose homeostasis. Enhances fatty acid oxidation and reduces body weight in mice through an increased production of leptin by the white adipose tissue (WAT). Inhibits Agxt2-mediated metabolism of dimethylarginines. Partial agonist of the glycine receptor (GlyR).
- SMILESCC(CN)C(O)=O
- Storage Instruction2°C to 8°C
- UNSPSC12352200