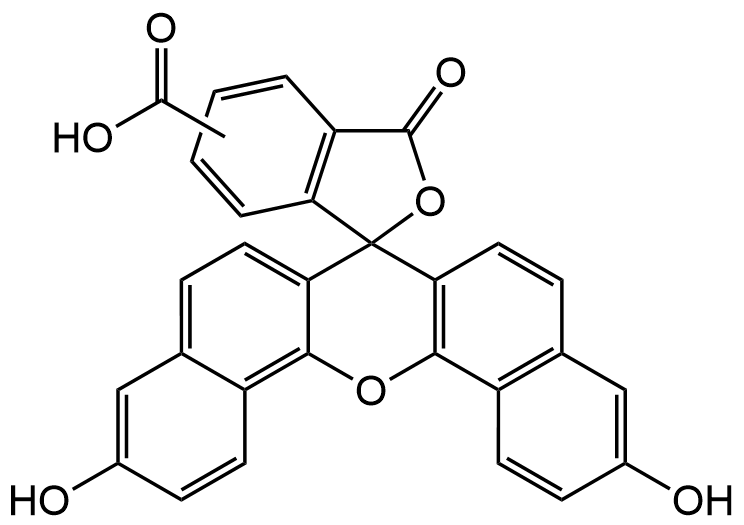
Chemical Structure
5(6)-Carboxynaphthofluorescein [128724-35-6]
CDX-C0047
CAS Number128724-35-6
Product group Chemicals
Estimated Purity>90%
Molecular Weight476.43
Overview
- SupplierChemodex
- Product Name5(6)-Carboxynaphthofluorescein [128724-35-6]
- Delivery Days Customer10
- CAS Number128724-35-6
- CertificationResearch Use Only
- Estimated Purity>90%
- Molecular FormulaC29H16O7
- Molecular Weight476.43
- Scientific Description5(6)-Carboxynaphthofluorescein (CNF) is a fluorescent NIR emitting pH indicator with red fluorescence at pH 9 (Spectral: Ex/Em: 598/668 nm). The fluorescence shifs to shorter wavelengths (Ex/Em: 512/567nm) at neutral or acidic pH environments. Spectral data: lambdaex=512nm; lambdaem=567nm (acid/neutral); lambdaex=598nm; lambdaem=668nm (basic). CNF shows good sensitivity in an alkaline pH range and it can be exploited in the construction of fiber-optic pH sensors. CNF has been used as an intravesicular pH probe for the colorimetric detection of activity, selectivity and cooperativity of ion channels. CNF has been used to design a luminescent probe for determination of pH, based on a Forster resonance energy transfer (FRET) system, combining a europium chelate as the donor and CNF as a pH sensitive acceptor. The FRET system enables referenced pH detection in an exceptional broad dynamic range from pH 3 to 9. - Chemical. CAS: 128724-35-6. Formula: C29H16O7. MW: 476.43. 5(6)-Carboxynaphthofluorescein (CNF) is a fluorescent NIR emitting pH indicator with red fluorescence at pH 9 (Spectral: Ex/Em: 598/668 nm). The fluorescence shifs to shorter wavelengths (Ex/Em: 512/567nm) at neutral or acidic pH environments. Spectral data: lambdaex=512nm; lambdaem=567nm (acid/neutral); lambdaex=598nm; lambdaem=668nm (basic). CNF shows good sensitivity in an alkaline pH range and it can be exploited in the construction of fiber-optic pH sensors. CNF has been used as an intravesicular pH probe for the colorimetric detection of activity, selectivity and cooperativity of ion channels. CNF has been used to design a luminescent probe for determination of pH, based on a Forster resonance energy transfer (FRET) system, combining a europium chelate as the donor and CNF as a pH sensitive acceptor. The FRET system enables referenced pH detection in an exceptional broad dynamic range from pH 3 to 9.
- SMILESOC(=O)c1ccc2c(c1)C(=O)OC23c4ccc5cc(O)ccc5c4Oc6c3ccc7cc(O)ccc67.OC(=O)c8ccc9C(=O)OC%10(c%11ccc%12cc(O)ccc%12c%11Oc%13c%10ccc%14cc(O)ccc%13%14)c9c8
- Storage InstructionRT
- UNSPSC12162000
