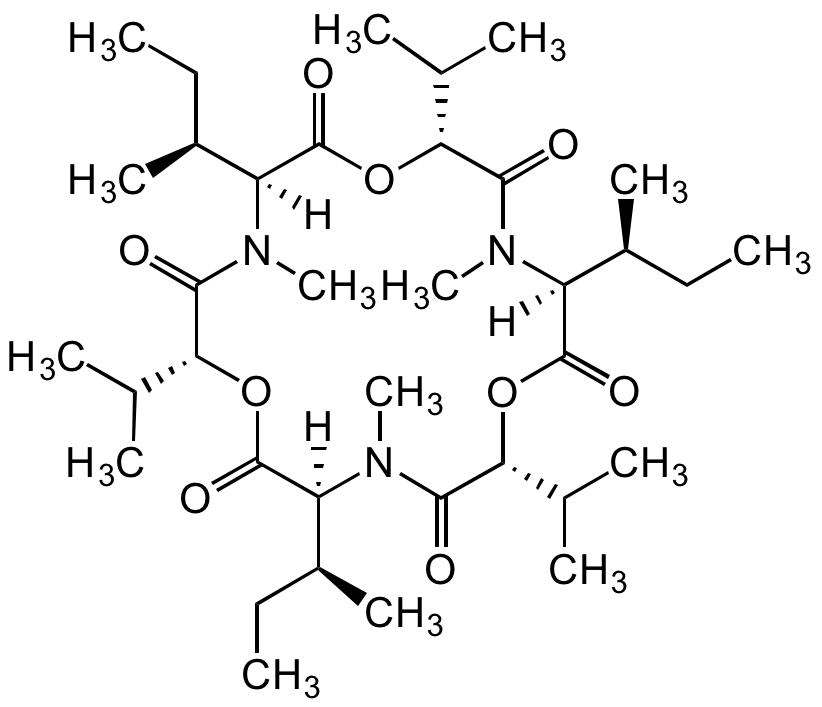
Chemical Structure
Enniatin A [2503-13-1]

AG-CN2-0477
Overview
- SupplierAdipoGen Life Sciences
- Product NameEnniatin A [2503-13-1]
- Delivery Days Customer10
- CAS Number2503-13-1
- CertificationResearch Use Only
- Estimated Purity>98%
- Hazard InformationWarning
- Molecular FormulaC36H63N3O9
- Molecular Weight681.9
- Scientific DescriptionChemical. CAS: 2503-13-1. Formula: C36H63N3O9. MW: 681.9. Isolated from fungus Fusarium sp. Cyclohexadepsipeptide mycotoxin. One of four major analogs in the enniatin complex. Commonly found food contaminant in cereals and their products. Ionophore antibiotic. Incorporation into the cell membrane forms dimeric structures that transport monovalent ions across the membrane (especially the mitochondrial membranes) affecting oxidative phosphorylation uncoupling. Anticancer compound. Triggers apoptosis in several cancer cell lines. Leads to loss of mitochondrial membrane potential and cell cycle arrest. Moderate inhibitor of ACAT (acylcoenzyme A:cholesterolacyl transferase). Shown to have a variety of other biological activities such as antifungal, anthelmintic, insecticidal, immunomodulatory and phytotoxic activity. - Cyclohexadepsipeptide mycotoxin. One of four major analogs in the enniatin complex. Commonly found food contaminant in cereals and their products. Ionophore antibiotic. Incorporation into the cell membrane forms dimeric structures that transport monovalent ions across the membrane (especially the mitochondrial membranes) affecting oxidative phosphorylation uncoupling. Anticancer compound. Triggers apoptosis in several cancer cell lines. Leads to loss of mitochondrial membrane potential and cell cycle arrest. Moderate inhibitor of ACAT (acylcoenzyme A:cholesterolacyl transferase). Shown to have a variety of other biological activities such as antifungal, anthelmintic, insecticidal, immunomodulatory and phytotoxic activity.
- SMILES[H][C@@]1([C@@H](C)CC)N(C)C(=O)[C@H](OC(=O)[C@]([H])([C@@H](C)CC)N(C)C(=O)[C@H](OC(=O)[C@]([H])([C@@H](C)CC)N(C)C(=O)[C@H](OC1=O)C(C)C)C(C)C)C(C)C
- Storage Instruction-20°C,2°C to 8°C
- UNSPSC12352200
References
- A comparison of beauvericin, enniatin and valinomycin as calcium transporting agents in liposomes and chromatophores: R.C. Prince, et al.; BBRC 59, 697 (1974)
- Inhibition of acyl-CoA: Cholesterol acyltransferase activity by cyclodepsipeptide antibiotics: H. Tomoda, et al.; J. Antibiot. (Tokyo) 45, 1626 (1992)
- Investigation of the electrophysiological properties of enniatins: M. Kamyar, et al.; Arch. Biochem. Biophys. 429, 215 (2004)
- Cytotoxicity of enniatins A, A1, B, B1, B2 and B3 from Fusarium avenaceum: L. Ivanova, et al.; Toxicon 47, 868 (2006)
- Enniatin exerts p53-dependent cytostatic and p53-independent cytotoxic activities against human cancer cells: R. Dornetshuber, et al.; Chem. Res. Toxicol. 20, 465 (2007)
- Emerging Fusarium-mycotoxins fusaproliferin, beauvericin, enniatins, and moniliformin - a review: M. Jestoi; Crit. Rev. Food Sci. Nutr. 48, 21 (2008)
- Oxidative stress and DNA interactions are not involved in Enniatin- and Beauvericin-mediated apoptosis induction: R. Dornetshuber, et al.; Mol. Nutr. Food Res. 53, 1112 (2009)
- Stimulation of erythrocyte phospholipid scrambling by enniatin A: K. Jilani, et al.; Mol. Nutr. Food Res. 55, 294 (2011)
- Toxigenicity of enniatins from Western Australian Fusarium species to brine shrimp (Artemia franciscana): D.C. Tan, et al.; Toxicon 57, 817 (2011)
- Revisiting the enniatins: a review of their isolation, biosynthesis, structure determination and biological activities: A.A. Sy-Cordero, et al.; J. Antibiot. 65, 541 (2012)
