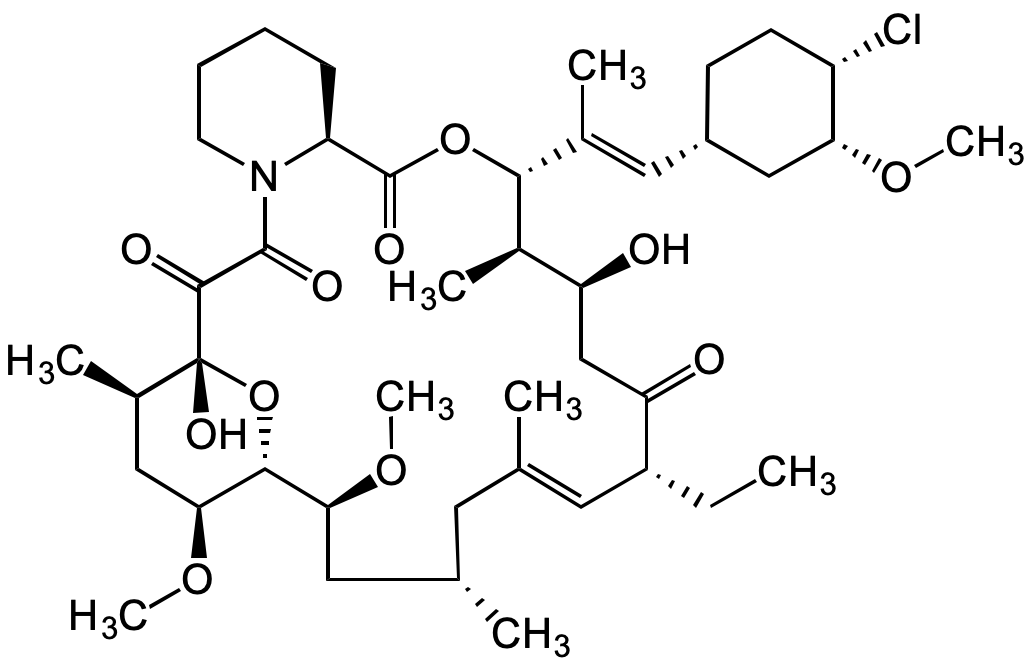
Chemical Structure
Pimecrolimus [137071-32-0] [137071-32-0]
CDX-P0598
CAS Number137071-32-0
Product group Chemicals
Estimated Purity>97%
Molecular Weight810.45
Overview
- SupplierChemodex
- Product NamePimecrolimus [137071-32-0] [137071-32-0]
- Delivery Days Customer10
- CAS Number137071-32-0
- CertificationResearch Use Only
- Estimated Purity>97%
- Molecular FormulaC43H68ClNO11
- Molecular Weight810.45
- Scientific DescriptionChemical. CAS: 137071-32-0. Formula: C43H68ClNO11. MW: 810.45. Synthetic. Pimecrolimus is a immunosuppressive natural ascomycin macrolactam that binds to macrophilin-12 (FKBP-12) and inhibits calcineurin as well as prolyl isomerase (rotamase). Presumably through these actions, it blocks the activation of T cells by allogeneic dendritic cells (IC50=0.55nM) without affecting dendritic cells. Moreover, pimecrolimus suppresses the generation of pro-inflammatory cytokines by T cells, the release of pre-formed inflammatory mediators from mast cells and the activation of eosinophils. These effects support the use of pimecrolimus in countering inflammatory skin diseases, such as atopic dermatitis (eczema) and psoriasis. - Pimecrolimus is a immunosuppressive natural ascomycin macrolactam that binds to macrophilin-12 (FKBP-12) and inhibits calcineurin as well as prolyl isomerase (rotamase). Presumably through these actions, it blocks the activation of T cells by allogeneic dendritic cells (IC50=0.55nM) without affecting dendritic cells. Moreover, pimecrolimus suppresses the generation of pro-inflammatory cytokines by T cells, the release of pre-formed inflammatory mediators from mast cells and the activation of eosinophils. These effects support the use of pimecrolimus in countering inflammatory skin diseases, such as atopic dermatitis (eczema) and psoriasis.
- SMILESCl[C@@H]1[C@@H](C[C@@H](CC1)/C=C(C)/[C@H]([C@H](C)[C@H](C2)O)OC([C@H](CCCC3)N3C(C([C@@](O)([C@H](C)C4)O[C@H]([C@H](C[C@@H](C)C/C(C)=C/[C@@H](CC)C2=O)OC)[C@H]4OC)=O)=O)=O)OC
- Storage Instruction2°C to 8°C
- UNSPSC12352200


![Pimecrolimus [137071-32-0] [137071-32-0]](https://www.targetmol.com/group3/M00/36/E3/CgoaEWayRUOEX8ZeAAAAAAvTUyo569.png)