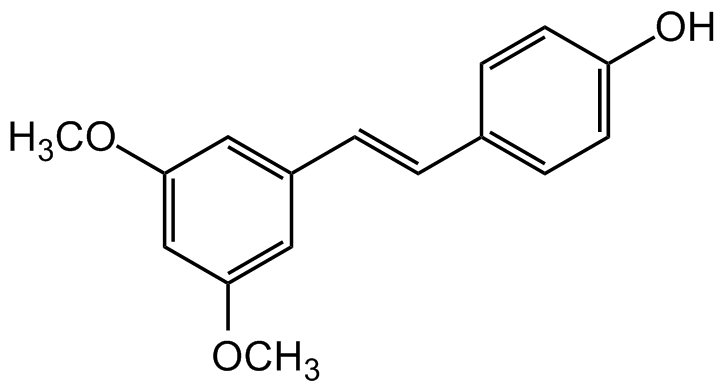
Chemical Structure
Pterostilbene [537-42-8]
CDX-P0234
CAS Number537-42-8
Product group Chemicals
Estimated Purity>97%
Molecular Weight256.3
Overview
- SupplierChemodex
- Product NamePterostilbene [537-42-8]
- Delivery Days Customer2
- CAS Number537-42-8
- CertificationResearch Use Only
- Estimated Purity>97%
- Hazard InformationDanger,Excepted quantity
- Molecular FormulaC16H16O3
- Molecular Weight256.3
- Scientific DescriptionCell permeable natural methoxylated analog of resveratrol. Antioxidant, antiproliferative, anti-inflammatory, anti-hyperglycemic and anti-diabetic agent. Induces apoptosis. Induces Autophagy. Pterostilbene is a naturally-occurring dimethyl ether analog of resveratrol that is abundant in blueberries. Like resveratrol, pterostilbene acts as a powerful antioxidant, suppresses the synthesis of prostaglandin E2 from lipopolysaccharide-stimulated human peripheral blood mononuclear cells (IC50 = 1.0 microM for pterostilbene, 3.2 microM for resveratrol), and inhibits cell proliferation (IC50 = ~60 microM for both compounds). Very potent inhibitor of cytochrome P450 1A1 (CYP1A1) with Ki=0.57microM. Pterostilbene blocks the activation of ERK1/2, p38 MAPK, and PI3K/Akt signaling pathways, reducing NF-kappaB and AP-1 transcriptional activation. Moderately inhibits COX-1 and COX-2 (IC50=19.8microM and 83.9microM). - Chemical. CAS: 537-42-8. Formula: C16H16O3. MW: 256.3. Synthetic. Cell permeable natural methoxylated analog of resveratrol. Antioxidant, antiproliferative, anti-inflammatory, anti-hyperglycemic and anti-diabetic agent. Induces apoptosis. Induces Autophagy. Pterostilbene is a naturally-occurring dimethyl ether analog of resveratrol that is abundant in blueberries. Like resveratrol, pterostilbene acts as a powerful antioxidant, suppresses the synthesis of prostaglandin E2 from lipopolysaccharide-stimulated human peripheral blood mononuclear cells (IC50 = 1.0 microM for pterostilbene, 3.2 microM for resveratrol), and inhibits cell proliferation (IC50 = ~60 microM for both compounds). Very potent inhibitor of cytochrome P450 1A1 (CYP1A1) with Ki=0.57microM. Pterostilbene blocks the activation of ERK1/2, p38 MAPK, and PI3K/Akt signaling pathways, reducing NF-kappaB and AP-1 transcriptional activation. Moderately inhibits COX-1 and COX-2 (IC50=19.8microM and 83.9microM).
- SMILESOC(C=C1)=CC=C1/C=C/C2=CC(OC)=CC(OC)=C2
- Storage Instruction2°C to 8°C
- UN NumberUN3077
- UNSPSC12352200