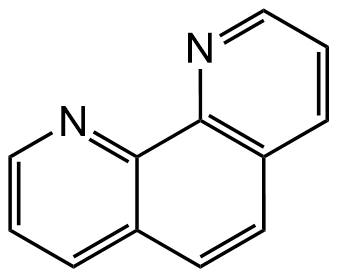
Chemical Structure
1,10-Phenanthroline [66-71-7]
CDX-P0153
CAS Number66-71-7
Product group Chemicals
Estimated Purity>98%
Molecular Weight180.21
Overview
- SupplierChemodex
- Product Name1,10-Phenanthroline [66-71-7]
- Delivery Days Customer10
- CAS Number66-71-7
- CertificationResearch Use Only
- Estimated Purity>98%
- Hazard InformationDanger,Excepted quantity
- Molecular FormulaC12H8N2
- Molecular Weight180.21
- Scientific Description1,10-Phenanthroline is a heterocyclic organic compound, which forms complexes with metal ions. It is a versatile ligand employed in the spectrophotometric determination of metals and photocatalytic reduction of carbon dioxide. The complexes that are build have wide applications in biochemistry, catalysis and analytical chemistry. 1,10-Phenanthroline is a ligand used in a mild, copper (II)-catalyzed cross-coupling of organoboronic acids and sulfinate salts leading to aryl- and alkenylsulfones. It is a conventional chelator to study its efficacy in Fentons reaction-luminol chemiluminescence system. 1,10-Phenanthroline enhances hPSC differentiation into cranial placode cells and is known as a non-specific matrix metalloproteinase (MMP) inhibitor. 1,10-Phenanthroline is also used as a building block for metallomacrocycles and other biological active compounds, such as anti-cancer agents. - Chemical. CAS: 66-71-7. Formula: C12H8N2. MW: 180.21. Soluble in DMSO (20mg/ml) or ethanol (20mg/ml). 1,10-Phenanthroline is a heterocyclic organic compound, which forms complexes with metal ions. It is a versatile ligand employed in the spectrophotometric determination of metals and photocatalytic reduction of carbon dioxide. The complexes that are build have wide applications in biochemistry, catalysis and analytical chemistry. 1,10-Phenanthroline is a ligand used in a mild, copper (II)-catalyzed cross-coupling of organoboronic acids and sulfinate salts leading to aryl- and alkenylsulfones. It is a conventional chelator to study its efficacy in Fentons reaction-luminol chemiluminescence system. 1,10-Phenanthroline enhances hPSC differentiation into cranial placode cells and is known as a non-specific matrix metalloproteinase (MMP) inhibitor. 1,10-Phenanthroline is also used as a building block for metallomacrocycles and other biological active compounds, such as anti-cancer agents.
- SMILESC12=CC=CN=C1C3=C(C=CC=N3)C=C2
- Storage InstructionRT
- UN NumberUN2811
- UNSPSC12162000

![o-Phenanthroline [66-71-7]](https://www.targetmol.com/group3/M00/37/29/CgoaEWaySRGELsbIAAAAABffhhM727.png)
