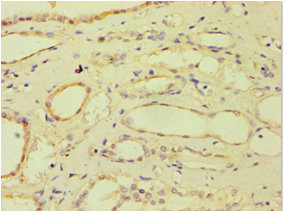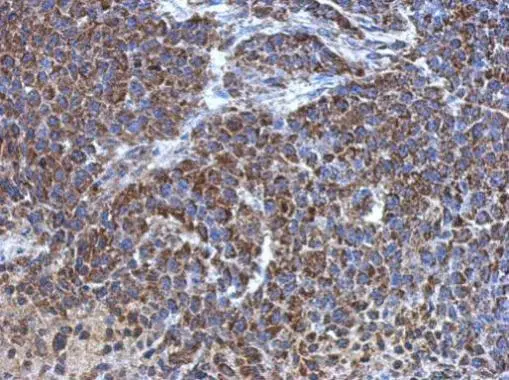
Immunohistochemistry of paraffin-embedded human breast cancer using CSB-PA002369ESR2HU at dilution of 1:100
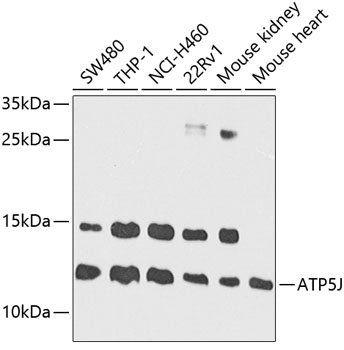
ATP5J Antibody
CSB-PA002369ESR2HU
ApplicationsELISA, ImmunoHistoChemistry
Product group Antibodies
ReactivityHuman
TargetATP5PF
Overview
- SupplierCusabio
- Product NameATP5J Antibody
- Delivery Days Customer20
- ApplicationsELISA, ImmunoHistoChemistry
- CertificationResearch Use Only
- ClonalityPolyclonal
- ConjugateUnconjugated
- Gene ID522
- Target nameATP5PF
- Target descriptionATP synthase peripheral stalk subunit F6
- Target synonymsATP5, ATP5A, ATP5J, ATPM, CF6, F6, ATP synthase peripheral stalk subunit F6, mitochondrial, ATP synthase subunit h, ATP synthase, H+ transporting, mitochondrial F0 complex, subunit F6, ATP synthase, H+ transporting, mitochondrial Fo complex subunit F6, ATP synthase-coupling factor 6, mitochondrial, ATPase subunit F6, coupling factor 6, mitochondrial ATP synthase, coupling factor 6, mitochondrial ATP synthase, subunit F6, mitochondrial ATPase coupling factor 6, proliferation-inducing protein 36
- HostRabbit
- IsotypeIgG
- Protein IDP18859
- Protein NameATP synthase peripheral stalk subunit F6, mitochondrial
- Scientific DescriptionMitochondrial membrane ATP synthase (F1F0 ATP synthase or Complex V) produces ATP from ADP in the presence of a proton gradient across the membrane which is generated by electron transport complexes of the respiratory chain. F-type ATPases consist of two structural domains, F1 - containing the extramembraneous catalytic core and F0 - containing the membrane proton channel, linked together by a central stalk and a peripheral stalk. During catalysis, ATP synthesis in the catalytic domain of F1 is coupled via a rotary mechanism of the central stalk subunits to proton translocation. Part of the complex F0 domain and the peripheric stalk, which acts as a stator to hold the catalytic alpha3beta3 subcomplex and subunit a/ATP6 static relative to the rotary elements. Also involved in the restoration of oligomycin-sensitive ATPase activity to depleted F1-F0 complexes.
- ReactivityHuman
- Storage Instruction-20°C or -80°C
- UNSPSC41116161