ELISA Kit for Endoglin (ENG)
SEA980HU
Product group Assays
Overview
- SupplierCloud-Clone Corp.
- Product NameELISA Kit for Endoglin (ENG)
- Delivery Days Customer12
- ApplicationsELISA
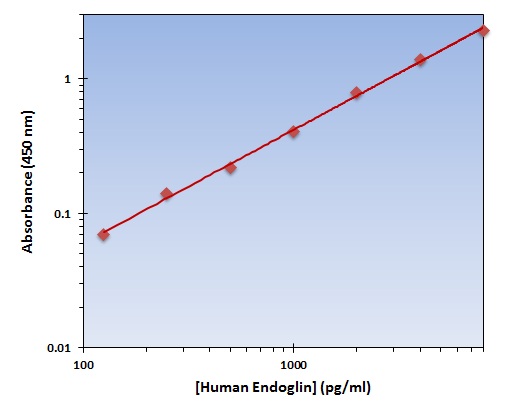
- Assay Detection Range0.156-10ng/mL
- Assay PrecisionIntra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Endoglin (ENG) were tested 20 times on one plate, respectively. Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level...
- Assay SensitivityThe minimum detectable dose of this kit is typically less than 0.061ng/mL
- Assay Test PrincipleThe test principle applied in this kit is Sandwich enzyme immunoassay. The microtiter plate provided in this kit has been pre-coated with an antibody specific to Endoglin (ENG). Standards or samples are then added to the appropriate microtiter plate...
- Assay Time3h
- CertificationResearch Use Only
- Protein IDP17813
- Protein NameEndoglin
- UNSPSC41116100
- SpeciesHuman
References
- Relationships between serum selenium and zinc concentrations versus profibrotic and proangiogenic cytokines (FGF-19 and endoglin) in patients with alcoholic liver cirrhosis. Prystupa A et al., 2017 Sep 21, Ann Agric Environ MedRead this paper
- Diagnostic and prognostic significance of serum soluble endoglin levels in preeclampsia and eclampsia. Sachan R et al., 2016, Adv Biomed ResRead this paper
- Impaired angiogenesis as a feature of digital ulcers in systemic sclerosis. Silva I et al., 2016 Jul, Clin RheumatolRead this paper
- Endothelial Dysfunction and Nailfold Videocapillaroscopy Pattern as Predictors of Digital Ulcers in Systemic Sclerosis: a Cohort Study and Review of the Literature. Silva I et al., 2015 Oct, Clin Rev Allergy ImmunolRead this paper