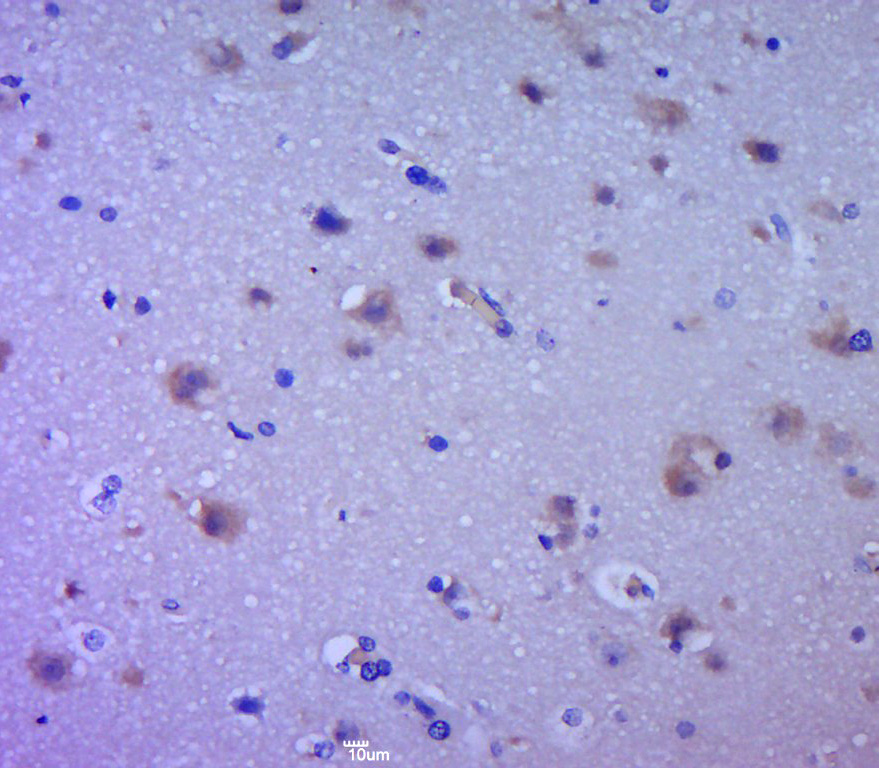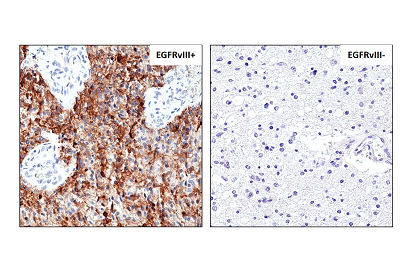
Sandwich ELISA, using RevMAb human IgG4 matched antibody pair, shows species reactivity to human only, and shows no cross-reactivity to monkey (Cyno or Rhesus), mouse IgG, rat IgG, or goat IgG.
anti-EGFRvIII (human), Rabbit Monoclonal (RM419)
REV-31-1305-00
ApplicationsFlow Cytometry, Western Blot, ELISA, ImmunoHistoChemistry
Product group Antibodies
ReactivityHuman
TargetEGFR
Overview
- SupplierRevMAb Biosciences
- Product Nameanti-EGFRvIII (human), Rabbit Monoclonal (RM419)
- Delivery Days Customer2
- ApplicationsFlow Cytometry, Western Blot, ELISA, ImmunoHistoChemistry
- CertificationResearch Use Only
- ClonalityMonoclonal
- Clone IDRM419
- Gene ID1956
- Target nameEGFR
- Target descriptionepidermal growth factor receptor
- Target synonymsERBB, ERBB1, ERRP, HER1, NISBD2, NNCIS, PIG61, mENA, epidermal growth factor receptor, EGFR vIII, avian erythroblastic leukemia viral (v-erb-b) oncogene homolog, cell growth inhibiting protein 40, cell proliferation-inducing protein 61, epidermal growth factor receptor tyrosine kinase domain, erb-b2 receptor tyrosine kinase 1, proto-oncogene c-ErbB-1, receptor tyrosine-protein kinase erbB-1
- HostRabbit
- IsotypeIgG
- Protein IDP00533
- Protein NameEpidermal growth factor receptor
- Scientific DescriptionEGFR (Epidermal growth factor receptor, HER1, ErbB1) belongs to the HER/ERbB family of proteins that includes three other receptor tyrosine kinases, ERbB2, ERbB3, ERbB4. EGFR is a transmembrane receptor and binding of its cognate ligands such as EGF (Epidermal Growth Factor) and TGF alpha (Transforming Growth Factor alpha) to the extracellular domain leads to EGFR dimerization followed by autophosphorylation of the tyrosine residues in the cytoplasmic domain (these include Y992, Y1045, Y1068, Y1148 and Y1173). Phosphorylation of EGF receptor (EGFR) at Tyr845 in the kinase domain is implicated in stabilizing the activation loop, maintaining the active state enzyme, and providing a binding surface for substrate proteins. The SH2 domain of PLCgamma binds at phospho-Tyr992, resulting in activation of PLCgamma-mediated downstream signaling. Phosphorylation of EGFR at Tyr1045 creates a major docking site for the adaptor protein c-Cbl, leading to receptor ubiquitination and degradation following EGFR activation. The GRB2 adaptor protein binds activated EGFR at phospho-Tyr1068. A pair of phosphorylated EGFR residues (Tyr1148 and Tyr1173) provide a docking site for the Shc scaffold protein, with both sites involved in MAP kinase signaling activation. Phosphorylation of EGFR at specific serine and threonine residues attenuates EGFR kinase activity. EGFR carboxy-terminal residues Ser1046 and Ser1047 are phosphorylated by CaM kinase II; mutation of either of these serines results in upregulated EGFR tyrosine autophosphorylation. EGFR activation signals multiple downstream signaling cascades such as the Ras - ERK, PI3K - Akt, Jak - STAT and PKC pathways that help in growth and proliferation of cells. Mutations in the EGFR gene (e.g. L858R) are associated with lung cancer and multiple alternatively spliced transcript variants encode different protein isoforms of EGFR have been found. The type III EGF deletion-mutant receptor (EGFRvIII) is the most common mutation and was first identified in primary human glioblastoma tumors. Increased production or activation of EGFR has been associated with poor prognosis in a variety of tumors. Moreover, EGFR overexpression is observed in tumors of the head and neck, brain, bladder, stomach, breast, lung, endometrium, cervix, vulva, ovary, esophagus, stomach and in squamous cell carcinoma. Deficient signaling of the EGFR and other receptor tyrosine kinases in humans is associated with diseases such as Alzheimers. - Recombinant Antibody. This antibody reacts to human Epidermal Growth Factor Receptor Variant III. No cross reactivity with wild type EGF receptor. Applications: IHC, WB, FACS, ELISA. Source: Rabbit. Liquid. 50% Glycerol/PBS with 1% BSA and 0.09% sodium azide. EGFR (Epidermal growth factor receptor, HER1, ErbB1) belongs to the HER/ERbB family of proteins that includes three other receptor tyrosine kinases, ERbB2, ERbB3, ERbB4. EGFR is a transmembrane receptor and binding of its cognate ligands such as EGF (Epidermal Growth Factor) and TGF alpha (Transforming Growth Factor alpha) to the extracellular domain leads to EGFR dimerization followed by autophosphorylation of the tyrosine residues in the cytoplasmic domain (these include Y992, Y1045, Y1068, Y1148 and Y1173). Phosphorylation of EGF receptor (EGFR) at Tyr845 in the kinase domain is implicated in stabilizing the activation loop, maintaining the active state enzyme, and providing a binding surface for substrate proteins. The SH2 domain of PLCgamma binds at phospho-Tyr992, resulting in activation of PLCgamma-mediated downstream signaling. Phosphorylation of EGFR at Tyr1045 creates a major docking site for the adaptor protein c-Cbl, leading to receptor ubiquitination and degradation following EGFR activation. The GRB2 adaptor protein binds activated EGFR at phospho-Tyr1068. A pair of phosphorylated EGFR residues (Tyr1148 and Tyr1173) provide a docking site for the Shc scaffold protein, with both sites involved in MAP kinase signaling activation. Phosphorylation of EGFR at specific serine and threonine residues attenuates EGFR kinase activity. EGFR carboxy-terminal residues Ser1046 and Ser1047 are phosphorylated by CaM kinase II; mutation of either of these serines results in upregulated EGFR tyrosine autophosphorylation. EGFR activation signals multiple downstream signaling cascades such as the Ras - ERK, PI3K - Akt, Jak - STAT and PKC pathways that help in growth and proliferation of cells. Mutations in the EGFR gene (e.g. L858R) are associated with lung cancer and multiple alternatively spliced transcript variants encode different protein isoforms of EGFR have been found. The type III EGF deletion-mutant receptor (EGFRvIII) is the most common mutation and was first identified in primary human glioblastoma tumors. Increased production or activation of EGFR has been associated with poor prognosis in a variety of tumors. Moreover, EGFR overexpression is observed in tumors of the head and neck, brain, bladder, stomach, breast, lung, endometrium, cervix, vulva, ovary, esophagus, stomach and in squamous cell carcinoma. Deficient signaling of the EGFR and other receptor tyrosine kinases in humans is associated with diseases such as Alzheimers.
- ReactivityHuman
- Storage Instruction-20°C,2°C to 8°C
- UNSPSC41116161